Alere™ HIV Combo
Features
-
A new standard in
HIV screeningAlere™ HIV Combo is an in vitro, visually read, qualitative lateral flow immunoassay for the simultaneous detection of free non immunocomplexed HIV-1 p24 antigen (Ag) and antibodies (Ab) to HIV-1 and HIV-2 in human blood. The test specimen can be serum, plasma, fingerstick or venous whole blood.
-
Any photos displayed are for illustrative purposes only.
-
Detection period (Weeks)
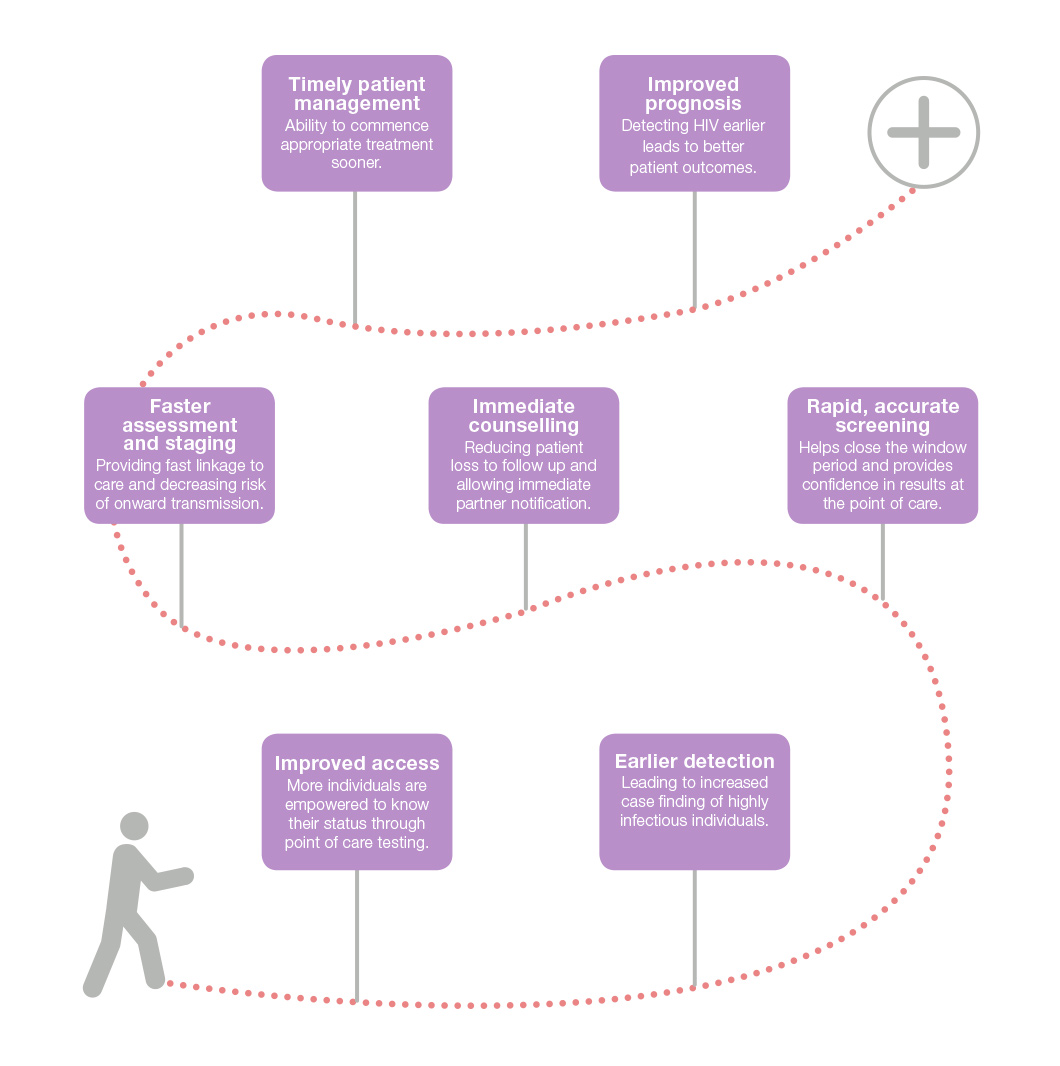
Alere™ HIV Combo detects more acute infections compared to 2nd and 3rd generation tests which helps close the window period and enables increased case finding at a time when individuals are highly infectious.
-
Simple Solution Complex Technology
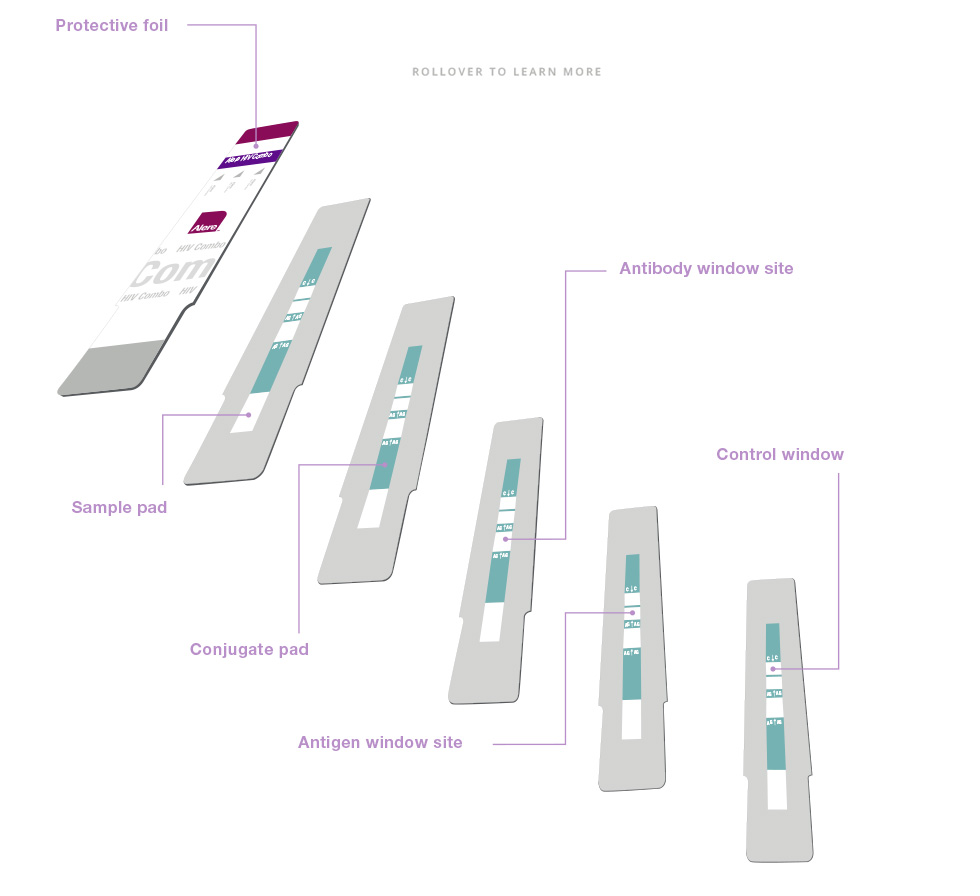 Preserves the assay reagent integrity for months at ambient temperatures (2-30°C)Preserves the assay reagent integrity for months at ambient temperatures (2-30°C)Serum, plasma or whole blood flows through the sample pad, where it activates proprietary reagents that trap red blood cells found in the sample.Serum, plasma or whole blood flows through the sample pad, where it activates proprietary reagents that trap red blood cells found in the sample.The specimen flows into the conjugate pad and the specific immunoreaction between the sample analyte and the assay specific reagents begins.The specimen flows into the conjugate pad and the specific immunoreaction between the sample analyte and the assay specific reagents begins.
Preserves the assay reagent integrity for months at ambient temperatures (2-30°C)Preserves the assay reagent integrity for months at ambient temperatures (2-30°C)Serum, plasma or whole blood flows through the sample pad, where it activates proprietary reagents that trap red blood cells found in the sample.Serum, plasma or whole blood flows through the sample pad, where it activates proprietary reagents that trap red blood cells found in the sample.The specimen flows into the conjugate pad and the specific immunoreaction between the sample analyte and the assay specific reagents begins.The specimen flows into the conjugate pad and the specific immunoreaction between the sample analyte and the assay specific reagents begins.As the specimen-conjugate mixture flows along the membrane strip, it passes through the patient window. If antibodies to HIV-1 and/or HIV-2 are present in the specimen, the antibodies bind to the reagents, forming one red bar at the Antibody window site.
If antibodies to HIV-1 and/or HIV-2 are absent it flows past the patient window, and no red bar is formed at the patient HIV Antibody window site.As the specimen-conjugate mixture flows along the membrane strip, it passes through the patient window. If antibodies to HIV-1 and/or HIV-2 are present in the specimen, the antibodies bind to the reagents, forming one red bar at the Antibody window site.
If antibodies to HIV-1 and/or HIV-2 are absent it flows past the patient window, and no red bar is formed at the patient HIV Antibody window site.If free non immunocomplexed HIV-1 p24 antigen is present in the specimen, the antigen binds to the reagents forming a red bar at the Antigen window site. If p24 antigen is not present the specimen-conjugate mixture flows past the patient window, and no red bar is formed at the Antigen window site.If free non immunocomplexed HIV-1 p24 antigen is present in the specimen, the antigen binds to the reagents forming a red bar at the Antigen window site. If p24 antigen is not present the specimen-conjugate mixture flows past the patient window, and no red bar is formed at the Antigen window site.As the specimen-conjugate mixture continues to flow along the membrane strip, it passes through the control window where other reagents are immobilized in a narrow bar. The control window bar functions as a procedural control to indicate assay validity and must always be present to correctly interpret the results. Approximately 20 minutes after the specimen is applied the test results can be read.As the specimen-conjugate mixture continues to flow along the membrane strip, it passes through the control window where other reagents are immobilized in a narrow bar. The control window bar functions as a procedural control to indicate assay validity and must always be present to correctly interpret the results. Approximately 20 minutes after the specimen is applied the test results can be read. -
A pathway to improved prognosis
with 4th generation point-of-care testing
-
Global Distribution of HIV-1 Subtypes and Recombinants2
HIV-1 antigens- HIV-1 group M subtype A, B, C, D, F, G, H and CRF01-AE, CRF02-AG, and HIV-1 group O are all detectable with Alere™ HIV Combo.3
HIV antibodies- HIV-1 group M subtype A, B, C, D, F, G, H, J, K, CRF01-AE, CRF02-AG, CRF03-AB, CRF05-DF, CRF09-A/U, CRF11-cpx, HIV-1 group O and HIV-2 are all detectable with Alere™ HIV Combo.3
-
Performance 3
Alere™ HIV Combo
Sensitivity*
100%
Specificity
99.72%
* Please refer to the Product Insert for analytical sensitivity of HIV-1 p24 antigen sensitivity.
References
- Edelstein, P. (2014) HIV Antibody Testing at HUP.
- Francine E. McCutchan, Henry M. Jackson Foundation (Rockville, Maryland). IAVI Report, August 2003 [Online] available at http://www.pbs.org/wgbh/pages/frontline/aids/atlas/clade.html
- Alere™ HIV Combo Product Insert