Alere™ q HIV-1/2 Detect
EID
-
Any photos displayed are for illustrative purposes only.
-
The Challenges of Early Infant Diagnosis
Current antibody point of care tests cannot discriminate between a mother’s and infant’s antibodies to detect if a newborn has become infected with Human Immunodeficiency Virus (HIV).
Virological testing of HIV nucleic acids is required for an accurate diagnosis in infants. This type of testing could only be performed in centralized laboratories with long lead times delaying results and the onset of critical treatment for children.
In the absence of timely HIV testing and antiretroviral therapy (ART) initiation, one third of infants living with HIV die before their first birthday, and half die before the age of two years1. Globally, the pace of scale-up of ART for children under 15 years is only half that of adults1.
Accurate diagnosis and access to antiretroviral therapy is a matter of life or death for infants.
-
“For the first time, we can provide HIV test results for infants to mothers on the same day that we conduct the test.”
Dr Landon Myer
-
-
Advancement in rapid HIV antibody tests has revolutionized access to testing and treatment in adult populations, however, to date we have been unable to provide our next generation with the same level of care.
According to a recent joint WHO/UNAIDS/UNICEF report only an estimated 28 percent of children born to HIV-positive mothers received an HIV test within the first two months of life1. Diagnosis of a HIV infection in infants remains a significant challenge due to the requirement of a virological test. In many settings this involves sending a sample to a centralized laboratory with a long turnaround time, or the service could simply not be available.
3.4 million children are living with HIV and only 34% of them are receiving ART1. We need to provide the next generation with better access to testing and treatment. The next generation of point of care diagnostics, the Alere™ q, is a revolutionary step in the right direction.
Alere™ is proud to be launching the first assay for the Alere™ q platform, the Alere™ q HIV-1/2 Detect for EID & Case ID, specifically to address the challenges of early infant diagnosis (EID) and adult diagnosis in the window period. This test will result in the same level of access for children to HIV testing and ART provided to adult patients.
Access to molecular testing can only reach scale if taken to the community rather than the community traveling to centralized laboratory locations.
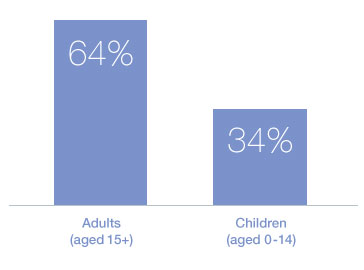
Percentage of adults (ages 15+) and children (aged 0-14) living with HIV receiving ART in low- and middle-income countries, 20122:
Data taken from United Nations Children’s Fund, Towards an AIDS-Free Generation – Children and AIDS: Sixth Stocktaking Report, 2013, UNICEF, New York, 2013. p11.2 -
Globally, the pace of scale-up of ART for children under 15 years is only half that of adults1.
-
References
- WHO/UNAIDS/UNICEF (2011) ‘ Global HIV/AIDS Response: Epidemic update and health sector progress towards Universal Access 2011).
- United Nations Children’s Fund, Towards an AIDS-Free Generation – Children and AIDS: Sixth Stocktaking Report, 2013, UNICEF, New York, 2013.